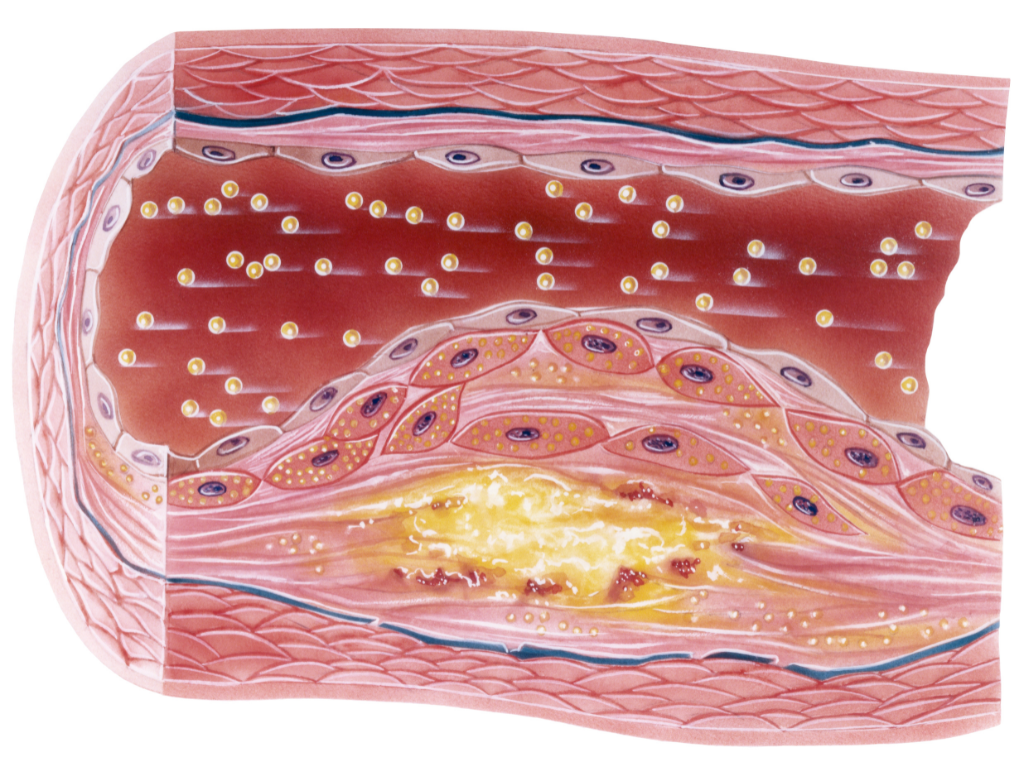
Due to the rapid progression of aging, as well as lifestyle and dietary imbalances, the number of patients with atherosclerosis is increasing. Atherosclerosis occurs in various parts of the body as a result of aging, dyslipidemia, diabetes, and other factors. In particular, peripheral arterial occlusive disease (ASO), characterized by the narrowing and impaired blood flow in the arteries of the legs, is predicted to continue rising. ASO is associated with complications and has been found to have a low long-term patient survival rate.
The mesenchymal stem cells used in this treatment have the ability to replicate themselves and differentiate into various cell types, contributing to blood improvement and vascular regeneration. Adipose-derived stem cells also have the property of secreting substances that suppress inflammation, which can help prevent worsening symptoms by reducing inflammation.
When administered through intravenous drip, the stem cells contribute to the repair of damaged tissues and promote tissue regeneration. It is believed that the secretion of cytokines and other substances by stem cells can suppress inflammation, while simultaneously producing substances that promote blood vessel regeneration, thus aiding in the recovery of blood flow in tissues affected by inadequate circulation.
Risks and side effects
When it comes to using autologous adipose-derived stem cells in stem cell therapy, it is generally believed that there are minimal direct side effects since the cells are derived from one's own body and cultured. Concerns regarding cancer development, which are often associated with induced pluripotent stem cells (iPSCs), are also not applicable in this case. Some rare reported side effects include minor complications such as bruising from the injection needle during fat tissue harvesting, localized pain around the site of fat extraction, and occasional cases of post-infusion fever. Additionally, there have been instances of individuals experiencing allergic reactions to anesthesia.
Process of regenerative therapy
A small amount of subcutaneous abdominal fat is harvested, and the adipose-derived stem cells contained within it are cultured and expanded to a quantity of over 100 million cells or more. These cells are then reintroduced into the body via intravenous drip to repair injured tissues and achieve rejuvenation.
We will listen to the patient's wishes and explain the purpose of regenerative medicine, treatment details, safety considerations, and other relevant information. Additionally, we will examine the patient's condition.

After obtaining consent from the patient, we will conduct tests to determine the presence or absence of specific viruses or bacteria related to infectious diseases. Based on the results, patients without any issues will proceed with the treatment.

We will perform fat harvesting in a clean and well-maintained procedure room. The harvesting method involves using a biopsy needle to extract subcutaneous fat. Local anesthesia will be applied, so you will likely feel minimal pain during the procedure. (The procedure typically takes about 20 minutes.)

- Side effects of fat tissue harvesting
After the fat tissue is harvested, there may be side effects such as swelling and bleeding at the incision site, internal bleeding in the harvested area, hyperpigmentation following internal bleeding, wound infection, and pain. If you experience any of these symptoms, our medical staff will address them appropriately.
The harvested fat is subjected to aseptic processing. Through enzymatic treatment, adipose-derived stem cells are separated from the fat and cultured in a culture vessel for approximately 2-3 weeks. It is important to note that no animal-derived components are used during the culturing process. Afterward, a certain quantity of cells is collected, and tests such as cell count and viability assessment, as well as quality examinations, are conducted. Once the confirmation of these results is completed, the cells are sent to the clinic for therapeutic use.

Stem cells are administered via intravenous drip over approximately one hour. In the case of multiple administrations, the cultured stem cells are frozen and stored in a cell processing facility. The standard protocol involves administering the cells every three months. Therefore, for subsequent administrations, there is no need for additional fat harvesting.
After administering the cultured stem cells, regular check-ups are conducted to monitor any changes in the patient's condition. Based on the results, adjustments are made to the dosage and frequency of administration. Additionally, advice is provided regarding home care, and support is provided to fulfill the patient's requests.

Treatment price
| 1 tmie | ¥2,900,000 (¥3,190,000 including tax) |
| 2 times | ¥4,900,000 (¥5,390,000 including tax) |
Individuals who are unable to receive treatment
- Individuals who do not understand the content of the informed consent document for this regenerative therapy.
- Individuals with hypersensitivity to the anesthesia used during fat harvesting.
- Individuals who may be pregnant.
- Individuals whom the treating physician deems unsuitable for the implementation of this regenerative therapy.
- Individuals under the age of 20 or over the age of 85.
- Individuals who tested positive for pathogenic microorganisms (such as HIV, syphilis) in the past 6 months.
| Both the patient and the physician must reach an agreement for stem cell therapy to be administered. Therefore, before the treatment, the physician will explain the "purpose, safety, risks, and benefits" of the treatment. Please carefully consider these factors and fully understand the treatment methods, expected outcomes, potential side effects, and any other explanations provided by the physician before giving your consent for the treatment. |
